 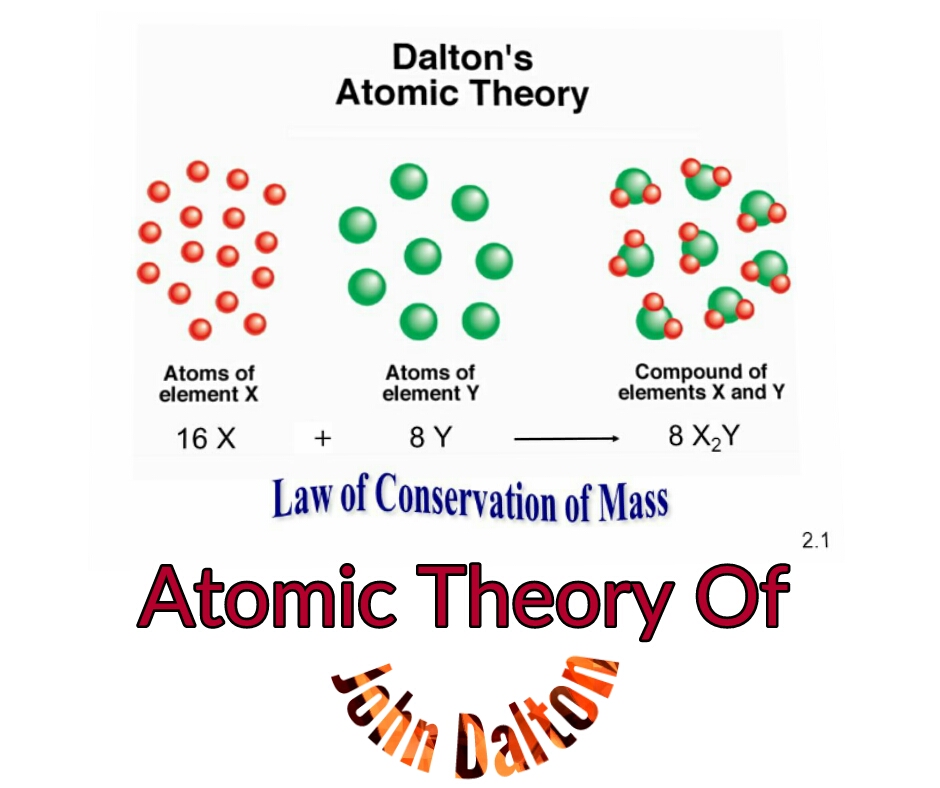
Instead, he focused upon determining the relative masses of each different kind of atom, a process that could be accomplished, he claimed, only by considering the number of atoms of each element present in different chemical compounds. 
His argument that each element had its own kind of atom was counterintuitive to those who believed that having so many different fundamental particles would destroy the simplicity of nature, but Dalton dismissed their objections as fanciful. Dalton claimed that atoms of different elements vary in size and mass, and indeed this claim is the cardinal feature of his atomic theory. 
Although this view was later shown to be erroneous, it served a useful purpose in allowing him to abolish the idea, held by many previous atomists from the Greek philosopher Democritus to the 18th-century mathematician and astronomer Ruggero Giuseppe Boscovich, that atoms of all kinds of matter are alike. This conceptualization explained why each gas in a mixture behaved independently. He based his theory of partial pressures on the idea that only like atoms in a mixture of gases repel one another, whereas unlike atoms appear to react indifferently toward each other. Attempts to trace precisely how Dalton developed this theory have proved futile even Dalton’s own recollections on the subject are incomplete. See how John Dalton built his atomic theory on principles laid out by Henry Cavendish and Joseph-Louis Proust See all videos for this articleīy far Dalton’s most influential work in chemistry was his atomic theory. 
SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.In the 5th Step➞ atoms CAN be divided further (3 subatomic particles: protons, electrons, and neutrons). In the 2nd Step ➞ atoms of a given element are NOT always identical (isotopes). Today, some 215 years after John Dalton proposed his Atomic Theory of Matter in 1808, there are just two modifications that must be made: Dalton’s Atomic Theory Modifications: = The Modifications of Dalton’s Atomic Theory #5 - Atoms cannot be divided any further.But the atoms themselves are not changed during the process. #4 - Chemical reactions involve the re-organization of atoms.A given compound always has the same relative numbers and types of atoms. #3 - Compounds are formed when atoms of different elements are combined.The atoms of different elements are different. #2 - The atoms of a given element are identical.#1 - Each element is composed of tiny indestructible particles called atoms.The five postulates below summarize Dalton’s Atomic Theory… Dalton’s Atomic Theory of Matter, and his 5 Postulates Dalton’s Atomic Theory and 5 Postulates 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
